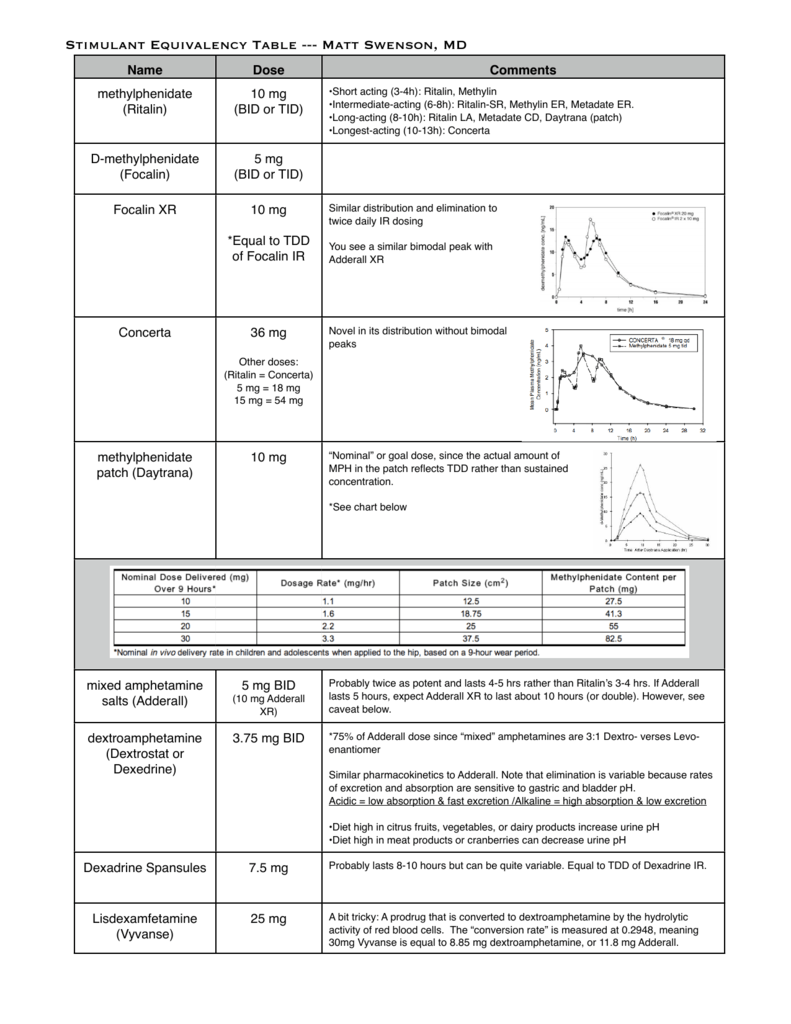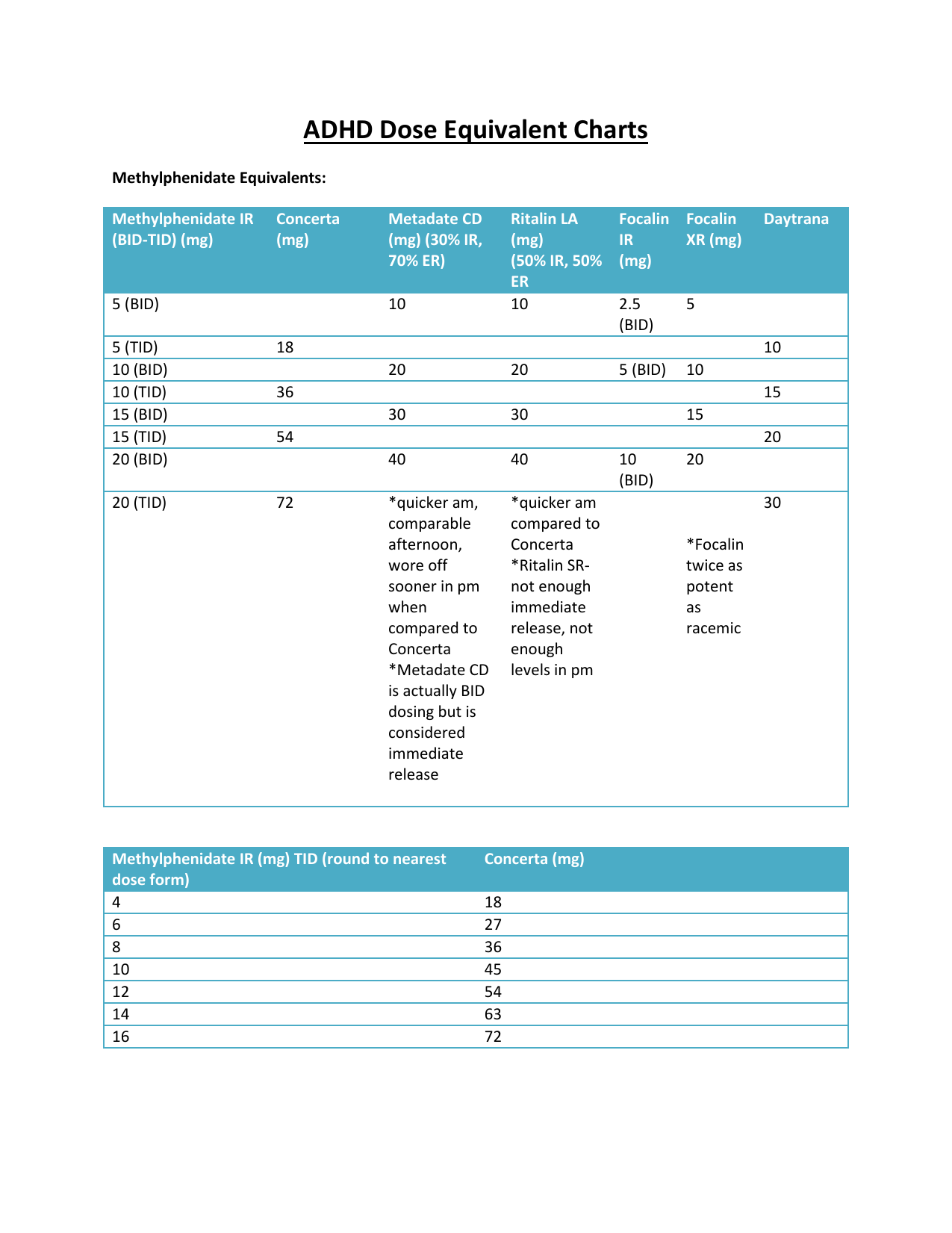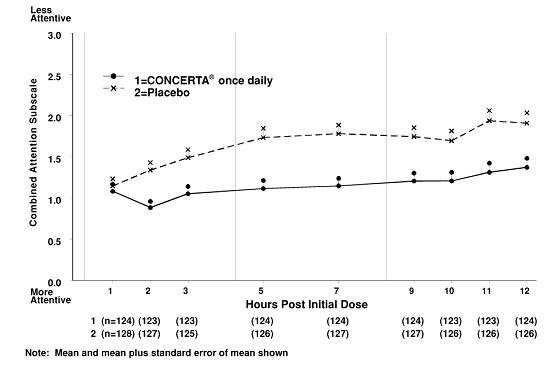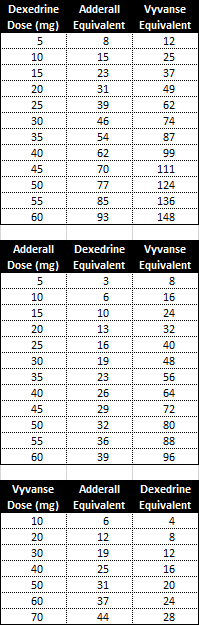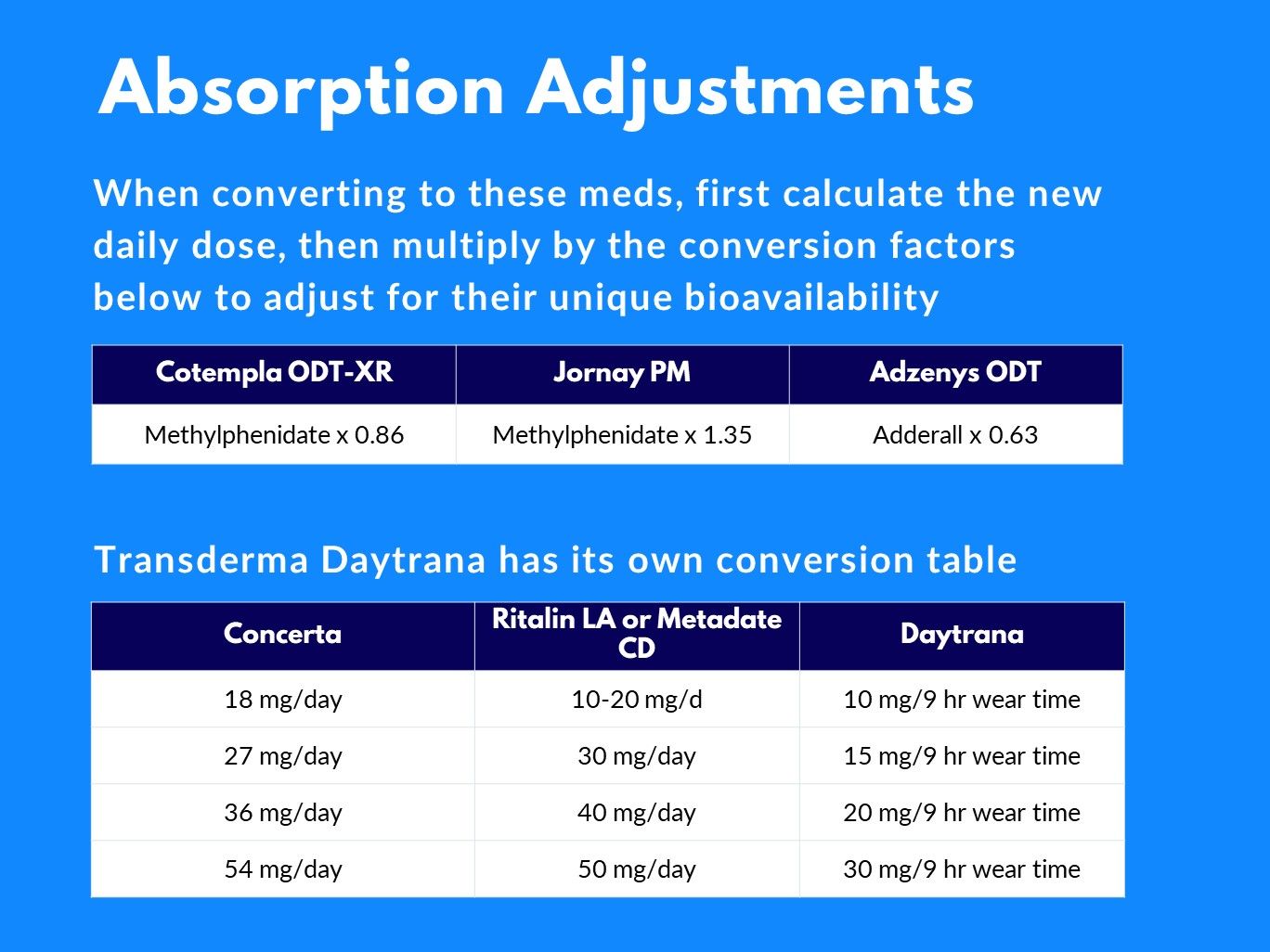
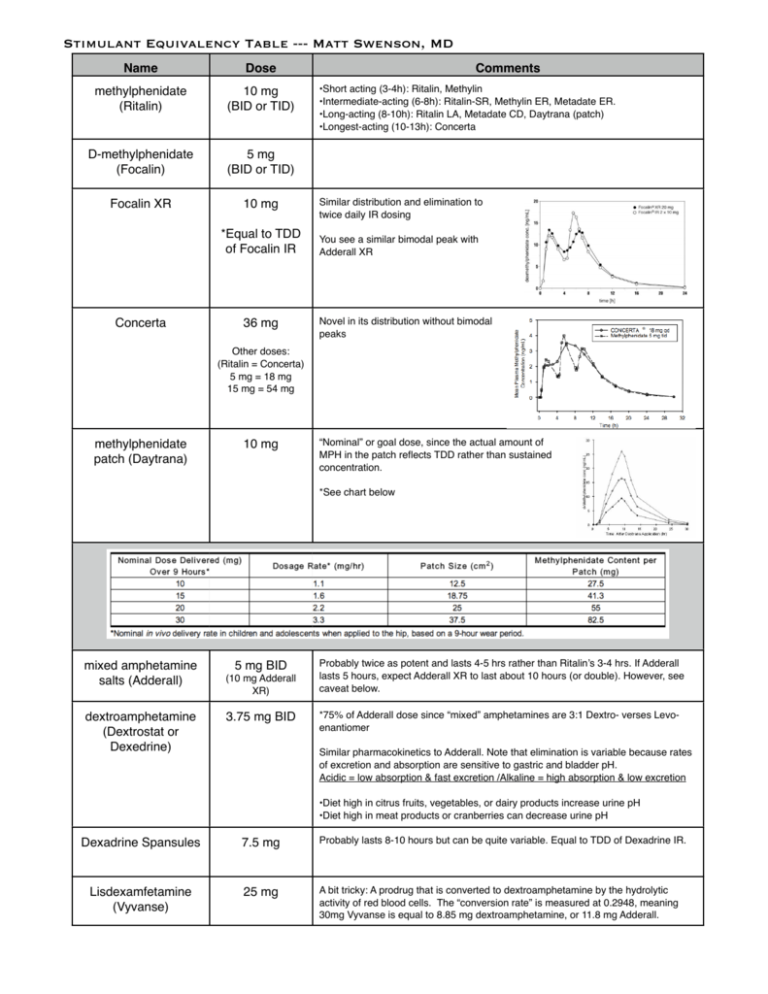
Stimulant Equivalency Table --- Matt Swenson, MD / stimulant-equivalency-table-matt-swenson-md.pdf / PDF4PRO
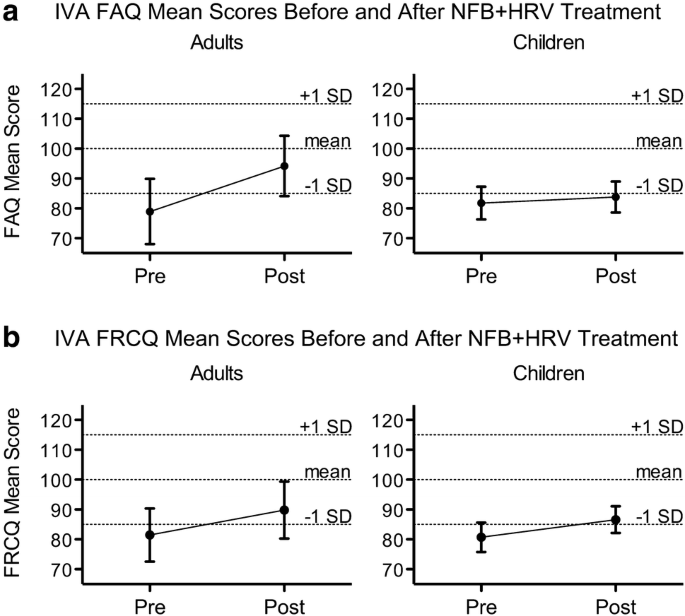
Z-Score Neurofeedback and Heart Rate Variability Training for Adults and Children with Symptoms of Attention-Deficit/Hyperactivity Disorder: A Retrospective Study | SpringerLink

Stimulant Equivalency Table --- Matt Swenson, MD / stimulant-equivalency-table-matt-swenson-md.pdf / PDF4PRO

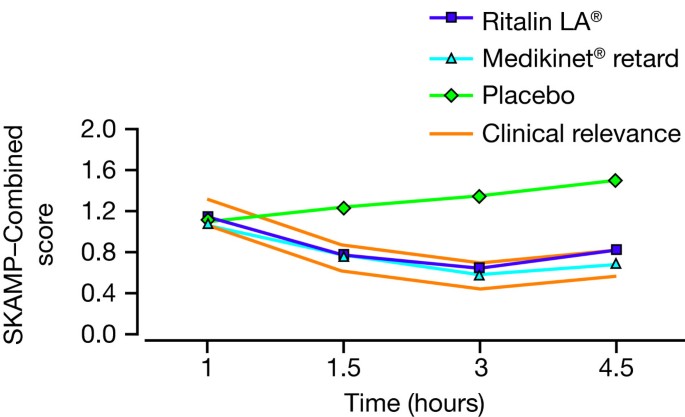
Comparative efficacy of Concerta®(18, 36 mg) and Ritalin LA®(20 mg)... | Download Scientific Diagram

Mixed amphetamine salts extended release: a clinical review of its use in the treatment of attention-deficit hyperactivity disorder | Future Neurology

MTS abrupt conversion study design. MTS = methylphenidate transdermal... | Download Scientific Diagram
Full article: Atomoxetine monotherapy compared with combination therapy for the treatment of ADHD: a retrospective chart review study

A Post Hoc Comparison of Prior ADHD Medication Dose and Optimized Delayed-release and Extended-release Methylphenidate Dose in a Pivotal Phase III Trial - Clinical Therapeutics

WO2015188092A1 - Methods for treating attention deficit hyperactivity disorder with methylphenidate - Google Patents
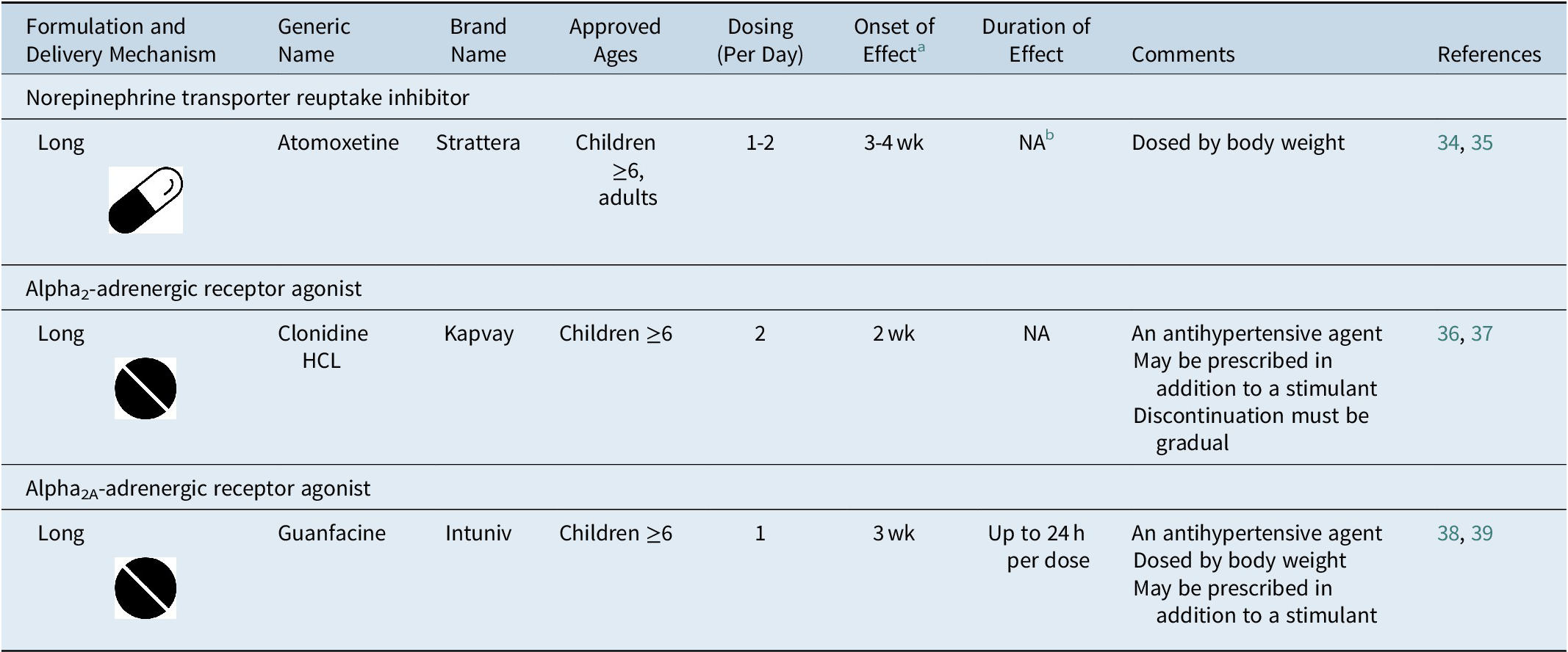
Individualization of attention-deficit/hyperactivity disorder treatment: pharmacotherapy considerations by age and co-occurring conditions | CNS Spectrums | Cambridge Core


/vyvanse-vs-adderall-4135491-Final-afc87beb39b14a13b68b409796ddbad3.png)